OneCell Diagnostics is now 1Cell.Ai
OncoIndx® TBx is an integrated NGS-based test designed to provide a complete genomic and transcriptomic profile of solid tumors.
It delivers clinically actionable insights that help oncologists personalize treatment strategies, identify resistance mechanisms, and improve patient outcomes.

The test integrates advanced AI interpretation, offering a single comprehensive report that includes mutations, fusions, copy number variations, and gene expression data — all aligned with the latest evidence-based therapies and clinical trials.
OncoIndx® TBx empowers clinicians by providing a unified, AI-powered view of tumor biology to guide personalized and targeted treatment decisions with confidence.
Comprehensive NGS panel covering 1000+ clinically relevant genes
Detects SNVs, INDELs, CNVs, and gene fusions
Includes HRD, MSI, TMB, and other key genomic signatures
Integrates RNA-based fusion detection for enhanced accuracy
AI-driven reporting through the iCARE™ platform
Simultaneous sequencing of DNA and RNA enables broad detection of variants, including single-nucleotide variants, indels, copy number variations, and fusions.
Delivers key biomarkers — HRD, MSI, LOH, and TMB — to assess genomic instability and predict response to immunotherapy or targeted therapy.
The iCARE™ AI engine consolidates variant data, literature evidence, and clinical guidelines into clear, actionable reports for therapy selection and trial matching.
Provides oncologists with precision insights to manage refractory, advanced, and treatment-resistant cancers.

Enables precise therapy selection for advanced or metastatic cancers
Provides clarity for patients with complex or refractory disease
Helps identify eligibility for targeted and immunotherapy trials
Comprehensive coverage of over 1000 genes across solid tumors
Dual DNA + RNA sequencing in a single test
Detects SNVs, INDELs, CNVs, fusions, and gene expression profiles
AI-powered analysis and reporting via the iCARE™ platform
Validated accuracy, sensitivity, and specificity (> 95 %)
Clear, clinically actionable insights aligned with NCCN and global guidelines
| Sample Type | FFPE tissue block (20–30% tumor content) |
| Sample Collection | 1 FFPE block (preferably <4 months old) |
| DNA/RNA Input Amount | >1 ng |
| Sequencing Platform | Illumina NGS (2 × 150 bp paired-end) |
| Sequencing Chemistry | Hybrid capture |
| Analysis Platform | iCARE™ AI-based analysis and dynamic reporting |
| Alteration | PPV | NPV | Accuracy | Specificity | Sensitivity |
|---|---|---|---|---|---|
| SNVs | 100 | 100 | 100 | 100 | 100 |
| Small INDELs | 100 | 94.03 | 97.40 | 100 | 95.60 |
| CNA | 100 | 100 | 100 | 100 | 100 |
| Fusions | 100 | 96.43 | 98.48 | 100 | 97.44 |
| RNA Fusion Assay (Validation) | PPV 78.6% | NPV 100% | Accuracy >95% within validated input range, precision >95%, LOD ≥20 ng RNA at ≥60× coverage | |||
| Cancer Type | Key Actionable Genes |
|---|---|
| Lung | EGFR, ALK, HRAS, NRAS, KRAS, BRAF, TERT, ROS1, MET, RET, NTRK1/2/3, ERBB2, PIK3CA |
| Breast | BRCA1, BRCA2, HER2, PIK3CA, MSH6, PMS2, NTRK1/2/3, MLH1, BRAF |
| Colorectal | BRAF, KRAS, NRAS, HRAS, ERBB2, MLH1, MSH2, MSH6, PMS2, NTRK1/2/3 |
| Prostate | BRCA1, BRCA2, MLH1, MSH2, MSH6, PMS2, AR, NTRK1/2/3 |
| Gastrointestinal | ALK, HRAS, HER2, NRAS, KRAS, BRAF, TERT, ROS1, MET, RET, NTRK1/2/3 |
| Thyroid | ALK, HRAS, NRAS, KRAS, BRAF, TERT, RET, NTRK1/2/3 |
| Bladder | FGFR2/3, NTRK1/2/3 |
| Melanoma | BRAF, KIT, ROS1, NTRK1/2/3 |
| Ovary | BRCA1, BRCA2, HER2, PMS2, MLH1, MSH2, MSH6, NTRK1/2/3 |
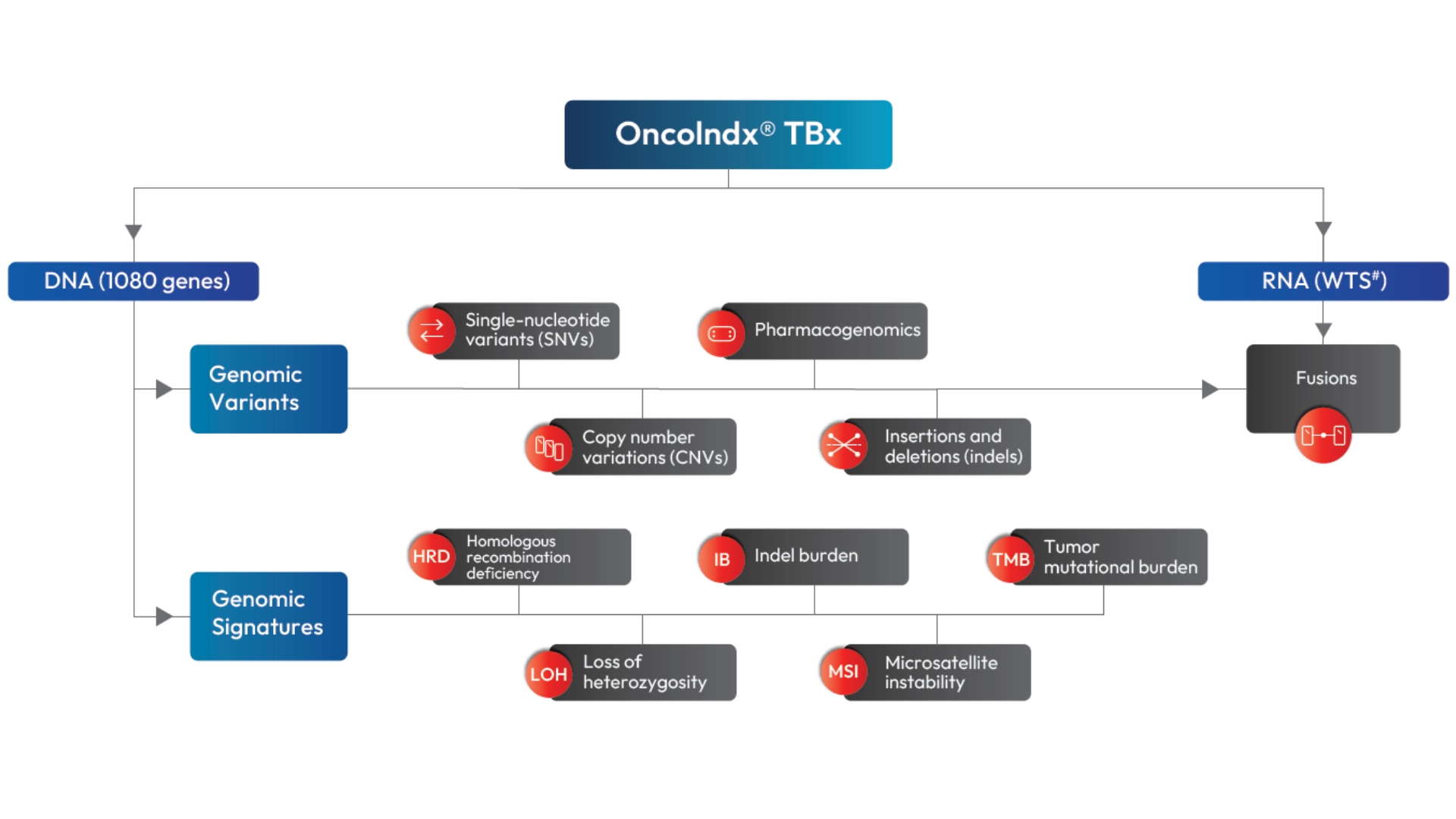
Integrated DNA + RNA sequencing for a complete molecular view
Identifies actionable mutations and resistance pathways
Dynamic reporting through the iCARE™ platform for clinical clarity
99 % on-target coverage and > 2000× mean sequencing depth
Supports precision oncology through informed, personalized therapy decisions