A Deep Dive into the Current Landscape of SERDs, ADCs, and Precision Oncology
The pace of breast cancer drug development has never been faster. From the practice-changing durability of oral selective estrogen receptor degraders (SERDs) to the increasingly crowded post-CDK4/6 landscape, the therapeutic arsenal continues to expand at a remarkable rate. Yet, a critical gap is widening: while therapies are becoming more sophisticated, our ability to reliably select the right patient for the right therapy at the right time is lagging.
An analysis of the recent clinical trial readouts, coupled with insights from ongoing field discussions, reveals a consistent pattern: therapeutic innovation is outpacing precision stratification.
Below is our in-depth assessment of where the field currently stands and where the most consequential gaps remain.
1. The Oral SERD Revolution: Breaking the 6-Month PFS Barrier
For the first time, the field has surpassed the long-standing ~6-month progression-free survival (PFS) ceiling in the second-line setting for patients with HR+/HER2-, ESR1-mutated metastatic breast cancer (mBC) following CDK4/6 and aromatase inhibitors.
- The New Standard: Oral SERDs are rapidly establishing themselves as practice-changing therapies. Elacestrant (Stemline) and the recently approved Imlunestrant (Lilly) are leading the charge.
- Emerging Powerhouses: Momentum continues to build as next-generation demonstrate increasing durability. Vepdegestrant (Arvinas/Pfizer) and Giredestrant (Genentech) have shown particularly compelling results, with Giredestrant demonstrating a PFS exceeding 10 months in ESR1-mutated patients.
- Moving to First-Line? Camizestrant (AstraZeneca) has shown a safety and efficacy profile that suggests potential utility in the first-line setting for ESR1-mutated populations.
The Takeaways:
- As these agents—along with contenders like Palazestrant—move earlier in the treatment sequence line of therapy, identifying ESR1 mutations accurately and tracking them longitudinally becomes critical
- There is growing recognition that longitudinal tissue biopsies are often impractical in patients with metastatic breast cancer (mBC). Although liquid biopsies based on circulating tumor DNA (ctDNA) have emerged as a less invasive alternative for precision oncology, they fail to detect clinically relevant genomic alterations in approximately 20–30% of patients, particularly those with low tumor shedding or non-shedding disease.
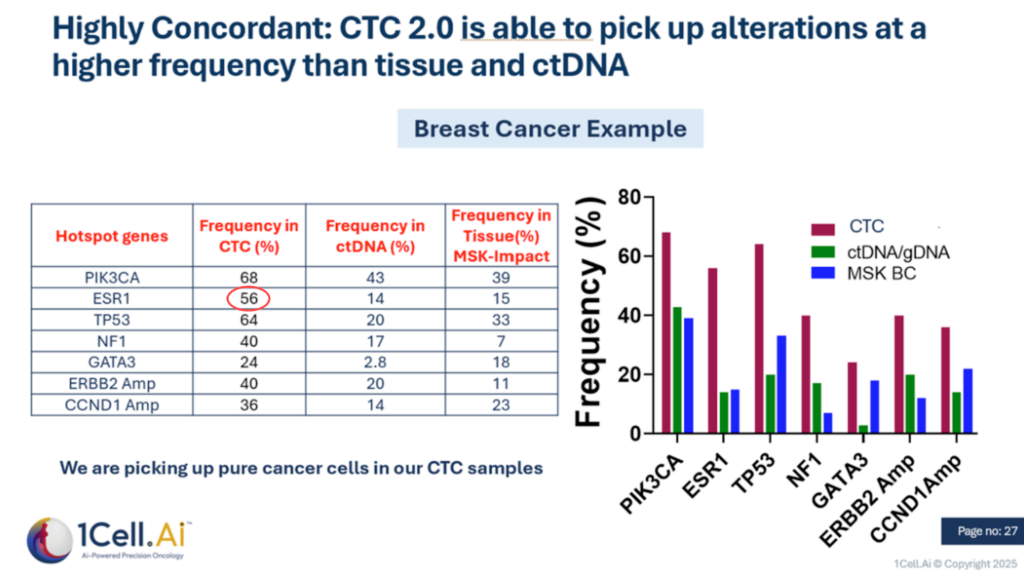
- As illustrated in Figure 1, next-generation circulating tumor cell (CTC 2.0) technologies can identify actionable molecular alterations in a substantial subset of these patients who are missed by both ctDNA-based assays and repeat tissue biopsies, thereby addressing a critical gap in current molecular profiling strategies.
Figure 1
2. The Post-CDK4/6 Kinase Race: Crowded and Complex
Resistance to CDK4/6 inhibitors is driving one of the most competitive races in oncology. The pipeline is flooded with next-generation kinase inhibitors targeting CDK2, CDK7, and KAT6, alongside renewed focus on the PAM pathway (PI3K/AKT/mTOR).
- The Players: Major pharmas (Pfizer, Novartis, AstraZeneca, Sanofi/Blueprint) and agile biotechs (BeOne, Avenzo, NiKang, Incyte) are all vying for dominance in this area.
- The Challenge: While companies like Celcuity are advancing PAM pathway activators, the fundamental question remains: Which specific resistance mechanism is driving the tumor?
- The Gap: Despite the volume of assets, it remains unclear if the current data is compelling enough to firmly establish these agents in the second-line setting without better biomarker stratification.
3. ADCs: Momentum Meets the “One-Size-Fits-All” Risk
Antibody-Drug Conjugates (ADCs) are redefining care across HER2-low, HER2+, and TNBC segments, but they are hitting a precision ceiling.
- HER2-Low & TNBC: 30% of the patients do not survive past 6 mo and will not receive 2L treatment; Trop2 ADCs from Gilead and Daiichi have shown promising data and are likely to move to 1L in combination with Pembro. However, no precision biomarker is being tested for Trop2 expression which may have changed with earlier lines of therapy.
- HER2+ mBC: Compelling data in Breast09 pivotal trial that shows Her2 ADC (Enhertu from Daiichi in combination with pertuzumab shows a 50% improvement in PFS; ADCs are likely to move to 1L similar to TNBC trend.
- The Stratification Flaw: Most of these programs rely on historical tissue status. They do not routinely retest for HER2 or Trop2 expression prior to treatment. Given that tumor biology evolves under therapeutic pressure, treating a patient based on a biopsy from months or years ago risks efficacy.
4. The Precision Paradox: Acknowledged but Unapplied
Industry dialogue reveals a troubling contradiction. Virtually every recent presentation concludes with a call for “better biomarkers.” Yet, field intelligence suggests a hesitation to implement them.
- The Reality: Discussions with field representatives from major players like Pfizer and Novartis confirm that many pipeline assets (both CDKs and ADCs) are currently taking a “non-precision” approach. While they acknowledge the risk, there is a perception that precision medicine adds commercial and operational complications.
- The Consequence: By avoiding deeper stratification beyond standard ESR1 or PAM mutations for SERDs and PI3K inhibitors or protein expression retesting for ADCs, we risk diluting the efficacy signals of potentially transformative drugs.
5. Liquid Biopsy 2.0: The Return of the Cell and Improved Sensitivity of ctDNA
Liquid biopsy is now standard of care, but we are reaching the limits of what ctDNA alone can do.
- The Standard: ctDNA testing is routinely used for ESR1 and PAM mutations, driven by the difficulty of obtaining tissue in mBC (accessible in only ~50% of patients).
- The Blind Spots: Recent data presented by the University of Michigan highlights that ctDNA often misses mutations—including ESR1 and PIK3CA—that are detectable in Circulating Tumor Cells (CTCs). See Figure 1 above.
- The Protein Problem: ctDNA cannot measure protein expression. For the booming ADC field, knowing the current expression level of HER2 or Trop2 on live cancer cells is vital.
- The Solution: 1Cell.Ai is demonstrating that CTCs can bridge this gap. By analyzing CTCs, we can capture both the mutational profile (often earlier than ctDNA) and the dynamic protein expression required for ADC selection.
6. Renewed interest in CTCs as a complement to ctDNA
As noted above, circulating tumor cells (CTCs) are now emerging as a viable and complementary modality to ctDNA for real-time biomarker profiling. Historically, CTC-based approaches were constrained by inefficient capture and limited downstream molecular characterization, resulting in false-negative rates of approximately 60–70%.
Recent advances in CTC enrichment, detection, and multi-omic analysis have overcome these limitations, enabling near-universal detection (~100% true positivity) in metastatic disease samples. As a result, CTC technologies have matured to a level of analytical robustness and reproducibility that supports their deployment in prospective clinical trials for longitudinal molecular profiling and therapeutic decision-making.
7. Multimodal AI emerging as a viable tool to analyze tissue images (e.g. H&E slides) to extract features and assess target biomarker expression:
There is emerging activity and innovation in the area of deploying multimodal AI to analyze tissue analysis all the way from improved diagnosis (e.g. highlighting areas of low expression of target) to predicting transcriptomic and protein expression signatures. In the poster presented by 1Cell.Ai, the company introduced its novel TRINITYAI multimodal AI offering and shared promising data to further solidify its accuracy and reproducibility. See attached poster here that shows that in this example, the Trinity AI shows that it has achieved xx% NPV and xx% specificity compared to Oncotype Dx’s recurrence score just by analyzing H&E images that have been archived.
Conclusion
The era of broad-stroke oncology is ending. To fully realize the promise of next-gen SERDs, CDK modulators, targeted agents against kinases and ADCs, we must move beyond static tissue and incomplete liquid biopsies. The future belongs to non-invasive, real-time, multi-omic stratification that sees the tumor as it is today.
Learn how 1Cell.Ai is solving the protein and mutation gap in liquid biopsy.